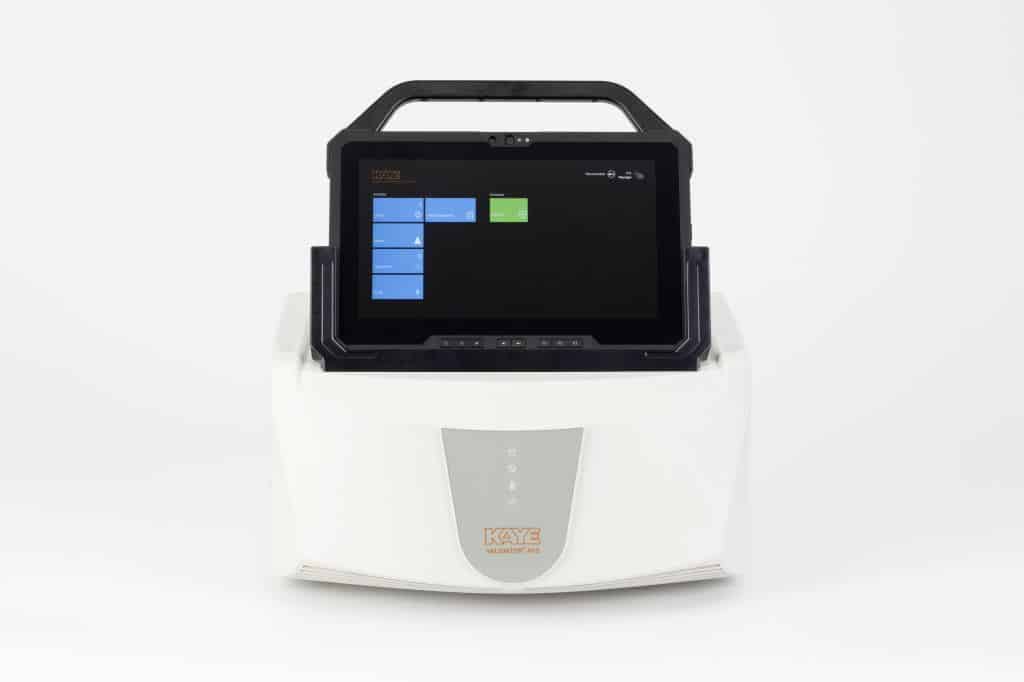
Kaye Temperature Validation System
Temperature Validation System: Kaye AVS
Discover the power of temperature validation systems in meeting and exceeding strict GMP compliance and regulations, these essential tools are ideal for pharmaceutical and biotechnology manufacturers. The AVS is a market-leading, reputable and advanced model for the completion of thermal validation. The system provides accurate and precise temperature and environmental measurements to comply with regulatory guidelines, GMP protocols and FDA approvals.
Temperature Validation Systems by Kaye
Kaye offers temperature validation systems to assist with temperature mapping, data logging, validation processes, qualification of equipment and regulatory compliance.
The Kaye AVS (advanced validation system) is a market-leading, highly accurate, sensor which meets stringent GMP requirements for documentation and reports.
In the pharmaceutical industry, ensuring the proper temperature and environmental conditions during the manufacturing and storage of products is critical for ensuring quality, safety and efficacy.
Thermal validation systems play a key role in achieving and documenting compliance with these processes. Find out how the Kaye AVS achieves this, as well as it's innovative features, below:
Please contact us for a quick quotation. The Kaye AVS is available for rental or purchase.
Complete the form below to access your free Kaye AVS user guide:

Meet the Kaye AVS
An Advanced Temperature Validation System
The Kaye AVS provides:
- Highly accurate sensor measurements
- Compliance with GMP requirements for calibration
- Fulfillment of traceability with national standards
- Compliant reports and auditable data
- Validated assessments of temperature and humidity equipment
To add, the temperature validation equipment from Kaye offers improved quality and usability, with this advanced temperature validation system, in particular, delivering user-friendly hardware features, an easy-to-configure data handling concept and an intuitive software interface.
The Kaye AVS operates with an integrated temperature validation system, providing improved data management as well as saving you valuable time and money through the simplified validation process.
The Kaye Validator AVS provides you with a completely new and upgraded validation concept. The AVS console will be the common platform for all validation products going forward. It is dedicated specifically to validation, removing worries about operating systems. The Software is preloaded and ready to go, with a customised interface for validation equipment, simplifying the validation process.
Applications of the Thermal Validation System
- Steam Sterilizers (Autoclaves)
- Dry Heat Sterilizers
- Steam in Place (SIP)
- Water Cascade/Fall Sterilizers
- Incubators
- Stability Chambers
- Freezers
- Freeze Dryer/Lyophilisation
- Vessels
- Enhanced data handling and redundancy (4-levels)
- Expanded input capacity, types, and range
- Improved scan time 2/sec for 48 inputs (EN554/ISO17665)
- Stand-alone operation with Console docking station
- Hardened, rugged Console pre-loaded with all Kaye software
- Easy-to-operate / state-of-the art intuitive user interface
- Ethernet/ WiFi/ Docking Station connections
- Asset centric concept for process equipment and Kaye hardware
- Console capable of running existing Kaye product software.
- Improved Reporting Analysis Tool –backward compatible
- Simplified validation (GAMP-5)
Please download our Kaye AVS brochure:
Expert Advice & Support Kaye AVS:
Our engineers provide independent, expert guidance to help you choose the right temperature validation system for your needs.
As an authorised Kaye partner in the UK & Ireland, we offer:
- Technical support and advice on Kaye AVS and other Kaye equipment
- UKAS-accredited calibration services
Contact us to learn more about the AVS thermal validation system or our full range of products and services.
Technical Specifications for Temperature Validation System:
| Analog Input | Up to 48 |
|---|---|
| Thermocouples | Type T, J, K,E,B,R,N,S: 0.1°C; T+ limited range 0.01°C resolution |
| Scanning speed | 36 channel in 1 sec and 48 channels in 2 Sec |
| Internal Memory | 4 gb for data collection |
| Input Impedence | 10KΩ. Source greater than 10KΩ produces open circuit indication |
| Common Mode Rejection | 160 db (8 inputs/sec) @ line frequency 145 db (12 inputs/sec) @ line frequency 140 db @ DC |
| Max. Common Mode Voltage | 100V pk ch-to-ch350V pk ch-to ch to frame ground |
| Normal Mode Rejection | 82 db @ 60 Hz (8 inputs/sec)69 db @ 60 Hz (12 inputs/sec) |
| Voltage Input | 0-10 VDC |
| Resolution | 1:72,000 |
| Voltage Input Accuracy | 30 days: ±(0.003% of reading + 2 counts + 4 microvolts) 1 year: ±(0.006% of reading + 2 counts + 4 microvolts) |
| Sensitivity | 0.5 microvolts/count on most sensitive range |
| Voltage Temp. Coef. | ±(0.1 microvolts + 0.001% reading)/°C |
| Compensator Temp. Coef. | ±0.01°C per °C |
| Input Terminal Temperature Non-uniformity | ±0.1°C from calibrated terminal |
| Input Ranges | -6 to 30mV, -12 to 60mV, -60 to 300mV, -2 to 10V |
| Environmental Ranges | Temperature: 0 to 50°C (32 to 122°F) Relative humidity: 95% non-condensing |
| Power | 90 to 250 VAC, 50/60 Hz |
| Fuse Rating | 4A Slow Blow |
| Size | 190H X 411W X 381 mm D (457 mm with SIM) 7.5 in H x 16.2 in W x 15 in D (18 in with SIM) |
| Weight | 10.60 kg (23.4 lbs) |
| Battery | Lithium ion with minimum 3 hours of battery backup |